IMPACT on Curbing Opioid Epidemic - RECENT REGULATORY TRENDS REFLECT - NABP
←
→
Page content transcription
If your browser does not render page correctly, please read the page content below
January 2022 | Volume 51 | Number 1 R E C E N T R E G U L AT O R Y T R E N D S R E F L E C T E-PRESCRIBING’S IMPACT on Curbing Opioid Epidemic
CONTENTS
05 11 01 Letter From the Chairperson
02 P
olicy Perspectives
Reverse Distribution: Serving Public
Health Under Range of State Rules
04 I nterview With a Board Inspector
Keith R. Bennett, PharmD, RPh
08 Association News
FDA Delays Enforcement Date for
Statutory Compounding Limit,
Extends Deadline for States’ MOU
Decisions
11 118th Annual Meeting
12 Proposed Resolutions Will
Be Distributed in February
13 NABP Announces 2022-
2023 Executive Committee
Openings; Elections to Take
Place During Annual Meeting
14 Interview With a Board Member
Ashlee Bow, PharmD, RPh, AAHIVP
16 State Board News
ansas Pharmacist Education
K
Program to Focus on
Feature News 118th Annual Meeting PDMP Utilization
Recent Regulatory Trends Join Fellow NABP Members
Reflect E-Prescribing’s Impact in the Valley of the Sun for 17 Professional Affairs Update
on Curbing Opioid Epidemic the 118th Annual Meeting NABP Joins Other Regulatory
Associations in Launching Opioid
Regulatory Collaborative
NABP Executive Committee
(ISSN 2472-6850 — print; ISSN 2472-6958 — Amy Sanchez Timothy D. Fensky Fred M. Weaver
online) is published 10 times a year by the National Publications and Editorial Manager Chairperson Member, District 4
Association of Boards of Pharmacy® (NABP®)
©2021 National Association of Boards of Pharmacy. Caroline D. Juran Shane R. Wendel
to educate, to inform, and to communicate the
All rights reserved. No part of this publication may President Member, District 5
objectives and programs of the Association and its
be reproduced in any manner without the written
65 member boards of pharmacy. Reginald B. “Reggie” Deborah C. Mack
permission of the executive director/secretary of the
Dilliard Member, District 6
The opinions and views expressed in this National Association of Boards of Pharmacy.
President-elect
publication do not necessarily reflect the official Nicole L. Chopski
NABP Mission Statement
views, opinions, or policies of NABP or any board Lenora S. Newsome Member, District 7
NABP is the independent, international, and
unless expressly so stated. The subscription rate is Treasurer
impartial Association that assists its member Kamlesh “Kam”
$70 per year. Bradley S. Hamilton Gandhi
boards in protecting the public health.
National Association of Boards of Pharmacy Member, District 1 Member, District 8
1600 Feehanville Drive, Mount Prospect, IL 60056 Tejal J. Patel
847/391-4406 | www.nabp.pharmacy Member, District 2 NABP Executive
help@nabp.pharmacy Committee elections
Jeffrey J. Mesaros are held each year
Lemrey “Al” Carter Member, District 3 at the Association’s
Executive Director/Secretary Annual Meeting.LETTER FROM THE CHAIRPERSON
Fellow Members,
Happy New Year! Like many people, I One major example of this is seen in the
have been using the transition into 2022 efforts of the boards of pharmacy to support
as an excuse to reflect on the successes of electronic prescribing practices. Because
the previous year and to identify areas of electronic prescriptions are considered
improvement. This helps me set goals for more secure and easier to track than paper
the coming year. This is also true for NABP. prescriptions, many states now require
We are now closing in on two years since the e-prescribing for at least some controlled
start of the COVID-19 pandemic. Much has substances. At the national level, recent
changed since the virus arrived in the United changes to Medicare requirements have also
States, but our focus on helping the boards made e-prescribing a requirement for certain
of pharmacy protect public health continues. types of medications. The cover story for
This has been reflected both in responses to this issue explores the differences among
the direct challenges created by COVID-19, state requirements and what impact they
as well as renewed efforts in other arenas, have had so far in improving public health.
many of which were already on our radar This issue also provides an update on
Timothy D. Fensky, long before the pandemic began. the Food and Drug Administration (FDA)
RPh, DPh, FACA Of course, one of these other arenas is memorandum of understanding (MOU)
NABP Chairperson the ongoing opioid crisis. As detailed in the with participating states. This important
October 2021 issue of Innovations, 2020 project focuses on a different area of public
saw a new record number of opioid overdose health that is also of great importance to the
fatalities – nearly a 30% increase from the boards of pharmacy – ensuring the safety
previous year. While this increase has largely of compounded medications. There have
been attributed to complications created by been several misconceptions about the data
the pandemic, as well as a continued increase sharing project, and my hope is that this
in the availability of illegally manufactured article will clarify how the MOU can help
synthetic opioids, prescription opioids boards address patient safety and improve
continue to be one avenue through which communication between FDA and the
many patients develop opioid use disorder boards of pharmacy.
(OUD). As has been widely reported As 2022 begins, I encourage each board
elsewhere, many of these patients turn to the and individual member to take advantage
black market for illicit opioids when they can of the transition as a time to reflect and set
no longer get prescription opioids. goals. Our profession is always evolving,
While there are limitations to what we and it is important that we take the time to
can do to prevent this, one thing that we think about what we can do to make sure
have been able to improve are practices that those changes have the best outcomes
related to the prescribing and dispensing in protecting the public health.
of prescription opioids. By preventing
overprescribing, we might be able to Sincerely,
reduce the number of people who develop
OUD. The data are still out as to whether
these efforts make a marked difference
in overall abuse, misuse, and diversion of
opioids. However, we can point to evidence
that shows prescription opioid abuse has Timothy D. Fensky, RPh, DPh, FACA
remained relatively steady, even during the NABP Chairperson
large increase in overall opioid overdoses over
the previous year.
And so, it remains important that we take
whatever steps we can to address the crisis.
JANUARY 2022 | 1POLICY PERSPECTIVES
Reverse Distribution: Serving Public Health Under Range
of State Rules
Reverse distribution is an important and uses USPS or a common carrier to ship
often overlooked component of the supply the product to its destruction facility, the
chain system. Reverse distributors are often reverse distributor will generally maintain
involved in the disposition or processing of title to such products even though possession
salable or nonsalable products received from an has been transferred to the shipping agent.
authorized trading partner. These products are This practice is important because state
then processed for credit or otherwise disposed licensure requirements often, but not
of and removed from distribution channels. always, depend on where in the distribution
The handling and removal of pharmaceutical chain a reverse distributor takes title and/or
products is a substantial public health matter possession of the pharmaceutical product.
and worth examining from a regulatory Generally speaking, if an out-of-state
context. reverse distributor picks up a pharmaceutical
Libby Baney, JD While some states specifically define product from a customer (and thus takes
Faegre Drinker Biddle & Reath LLP
reverse distributors and what constitutes title and possession of the pharmaceutical
reverse distribution, many other states product) to transport the product to
either include reverse distributors under its destruction facility, most states have
the umbrella of wholesale distributors or determined that a nonresident license in
simply do not address reverse distributors that nonresident state is required. However,
in the statute or regulations. Where there if the customer (eg, a retail pharmacy),
are regulations, they are often vague or rather than the reverse distributor, ships
inconclusive and require outreach to or transports the pharmaceutical product
regulators at the applicable governing body to the reverse distributor’s destruction
for clarification. Periodically, these regulators facility and the reverse distributor does not
interpret and apply their state regulations actually take possession of the product,
differently, leading to inconsistent guidance then the reverse distributor will generally
for reverse distributors. not be required to hold a nonresident
Jay A. Warmuth, JD
Faegre Drinker Biddle & Reath LLP
license in the customer’s state.
Reverse Distribution Process and If reverse distribution services are
Regulatory Common Ground interrupted, this can lead to major
Oftentimes, a reverse distributor receives disruption for retail and hospital
unwanted, unusable, or outdated pharmacy customers who must remove
pharmaceuticals from a pharmacy or other the pharmaceutical product from their
facility that ships the product(s) directly to premises. Given the diversion risks when
the reverse distributor. In other cases, the handling waste that involves controlled
reverse distributor will pick up and transport, substances (CS) and public health concerns
or arrange for the pickup and transport, of when handling pharmaceutial waste,
the unwanted pharmaceutical products from it is important that pharmacies utilize
the customer. In these situations, the reverse specialized, credible reverse distribution
distributor will either physically transport vendors to remove and dispose of the
the pharmaceutical products itself or ship waste in a timely and compliant manner.
Jonathan A. Keller, PharmD, JD, RP
Faegre Drinker Biddle & Reath LLP the products to its destruction facility via
the United States Postal Service (USPS) or Reverse Distributor Regulations
a common carrier. During this process, the Vary Among States
reverse distributor typically takes title and For the vast majority of states, reverse
possession of the pharmaceutical products distributors are regulated by the state
when the products are picked up from the board of pharmacy. In a handful of states,
customer. Even if the reverse distributor however, regulatory agencies other than the
2 | JANUARY 2022POLICY PERSPECTIVES
state board of pharmacy exercise regulatory questions or other guidance documents perform reverse distribution activities generally,
authority over reverse distributors. For regarding licensure of reverse distributors. but do require the reverse distributor to hold a
example, reverse distributors are overseen by While in other states, a board’s meeting CSR in order to handle CS. In these situations,
the Department of Health in the District minutes or wholesale distributor license the reverse distributor is typically required to
of Columbia and Washington State; by the application may include insights to help first obtain its state distributor license before it
Department of Consumer Protection in determine whether a reverse distributor is can apply for and obtain its CSR. For example,
Connecticut; by the Department of State required to hold a license in a specific state. the Hawaii State Board of Pharmacy does
in Delaware; by the Board of Drug and Once it is determined that a reverse not license nonresident reverse distributors;
Device Distributors in Louisiana; by the distributor should hold a license in a however, the State of Hawaii Department of
Department of Agriculture & Consumer particular state, there are oftentimes Public Safety, Narcotics Enforcement Division
Services in North Carolina; and by the additional considerations – one of which is has indicated that a nonresident reverse
Department of State Health Services in Texas. Drug Distributor Accreditation from NABP. distributor collecting CS within Hawaii is
Drug Distributor Accreditation indicates required to hold a CSR even though the Board
The Landscape of State whether a particular wholesaler meets certain of Pharmacy has indicated that its laws and
Licensing Requirements minimum compliance standards established regulations do not address reverse distributors.
A significant concern of regulators and reverse by NABP. Drug Distributor Accreditation is Similarly, a nonresident reverse distributor
distributors is the ambiguity surrounding a requirement for licensure in Indiana, Iowa, would not be required to hold a license from
whether a reverse distributor is required to be North Dakota, and Wyoming. the New York State Board of Pharmacy if
licensed in a particular state. Not all states license it only sends product out of New York. But
reverse distributors. For example, the Alaska Adding Complexity: New York’s Bureau of Narcotic Enforcement
Board of Pharmacy does not require a reverse Reverse Distribution of requires a reverse distributor to hold a CSR
distributor to hold a license in Alaska so long Controlled Substances to distribute or handle CS within New York.
as the reverse distributor does not resell the If a reverse distributor will be engaging in In both examples, the reverse distributor does
collected product. This appears to apply to both the reverse distribution of CS, there are not need to hold a distributor license from the
resident and nonresident reverse distributors. additional considerations to keep in mind. board of pharmacy but may need to obtain
Other states have taken a narrower In addition to holding the requisite CS one nonetheless in order to receive a CSR
approach and do not require reverse registration certificate from Drug Enforcement to handle or distribute CS in those states.
distributors to hold a nonresident license in Administration, the applicable state CS laws The regulation of reverse distribution of
certain circumstances. For instance, Idaho must also be reviewed. Some states will issue pharmaceuticals is complex and varied across
and Hawaii have indicated that they do not a separate state-level CS registration (CSR). the states. When developing regulation or
require nonresident reverse distributors to Other states will simply indicate on the reverse policies, or statutory language, boards of
hold a license to conduct business in their distributor’s underlying state license whether pharmacy may want to consider several
respective states. Other states have taken such entity is permitted to handle CS. questions. Is “reverse distributor” defined in
a different approach and tie nonresident Another consideration regarding CS is to the state’s law, or does the definition need to
licensure to other criteria. For example, a understand which state agency is responsible be included in regulatory language or board
reverse distributor not physically located for issuing the CSR. For example, the Iowa guidance documents? What reverse distributor
in Pennsylvania, but performing reverse Board of Pharmacy and Michigan Board of business activities need to be accounted for?
distribution services in Pennsylvania, is only Pharmacy are responsible for issuing both Are resident and nonresident state licensure
required to hold a nonresident license if a distributor license and a separate CSR to requirements clear? Do the regulations address
either (i) it has sales representatives physically reverse distributors handling CS in their the proper handling of CS to prevent diversion?
working or operating in Pennsylvania, or applicable jurisdiction. However, there are Do the regulations address hazardous waste
(ii) the reverse distributor is shipping or several states that have separate agencies disposal and compliance with Environmental
receiving product containing a list I chemical governing CSR licensure requirements. For Protection Agency requirements?
from a facility located in Pennsylvania. instance, the South Dakota State Board This article was written by Libby Baney, JD;
In some states, guidance on reverse of Pharmacy is the agency responsible for Jay A. Warmuth, JD; and Jonathan A. Keller,
distributor licensure may be provided issuing a distributor permit to a reverse PharmD, JD, RPh, with Faegre Drinker
in documents or policy other than distributor, but it is the South Dakota Biddle & Reath LLP. Please note, the opinions
the applicable statute and regulations, Department of Health that is responsible and views expressed by Faegre Drinker Biddle
especially if they are silent regarding reverse for issuing a CSR to a reverse distributor. & Reath do not necessarily reflect the official
distributors. For instance, some state boards Some states do not require a reverse views, opinions, or policies of NABP or any
of pharmacy have published frequently asked distributor to hold a distributor license to member board unless expressly stated.
JANUARY 2022 | 3INTERVIEW WITH A BOARD INSPECTOR
Keith R. Bennett, PharmD, RPh
Chief Inspector/Compliance Officer, Wyoming State
Board of Pharmacy
How long have you been an reference in the Board’s rules. At that time,
inspector for the Board? it was anticipated that the 2019 version of
I have been serving as an inspector for the USP and General Chapter
Wyoming State Board of Pharmacy since would become official and eventually take
September 2019. Prior to joining the Board, its place. Many sterile compounders in the
I worked as a contract pharmacist for the state were either planning or undergoing a
Civilian Health and Medical Program of remodel to come into compliance with the
the Department of Veterans Affairs. Before new chapters. The Board had also been in the
pursuing pharmacy as a career, I served on process of educating licensees on USP
active duty in the United States Air Force, standards in preparation of the new chapters
and I continue to serve in the Wyoming Air becoming official. Within my first seven
Wyoming State National Guard. months, the new chapters were remanded
Board of Pharmacy and USP became informational. As a
What tools or skills are a must-have result, the Board approved an emergency rule
in a pharmacy inspector’s toolkit? adopting the 2008 version of USP by
Number of Board The Wyoming Pharmacy Act Rules and reference. In May 2021, the Board approved
Members
Regulations, and other applicable federal and new rules for sterile compounding, and I have
5 pharmacist members,
1 public member, state laws that govern the practice of pharmacy been addressing issues and confusion to help
1 physician, 1 dentist are the most important “tools” I have as bring sterile compounders into compliance.
or veterinarian, and an inspector. Because policies, procedures,
1 pharmacy technician and processes can vary from pharmacy to Is there an inspection experience
pharmacy, a thorough understanding of that you found particularly
Number of pharmacy law is necessary to ensure accurate interesting, egregious, or unusual?
Compliance and consistent observations across inspections. The most egregious case I investigated
Officers/Inspectors Effective verbal and written communication involved a veterinary wholesaler that was
2 are also essential. When used effectively, repackaging and relabeling prescription
these skills foster positive relationships with drugs for use in ornamental fish, and then
licensees based on mutual respect as well distributing those products in other states
Rules & Regulations
Established by as allow an inspector to apply knowledge without the appropriate licenses and/or
State Board of of pharmacy law in a constructive manner. registrations. This case required thorough
Pharmacy These skills also promote an increased state research and understanding of federal law as
of compliance over time and an overall well as other states’ laws requiring licensure
positive perception of the Board. to engage in wholesale distribution. The
Number of Attention to detail is another important Board concluded that the distributor violated
Pharmacist Licensees skill. An inspector can be presented with the Wyoming Pharmacy Act by selling
1,437 various distractions and interruptions adulterated drugs, selling misbranded drugs,
throughout the inspection process. This can failing to maintain records, and engaging
lead to a pattern of inconsistent observations in unlicensed practice. The owner and
Number of over time, thereby reducing the effectiveness company were prohibited from renewing,
Pharmacies
of the inspection program. reinstating, or obtaining any license from
157
the Board for 10 years and will have to pay
What are some common issues that an administrative penalty of $500,000 for
you have witnessed and addressed any future attempt to renew, reinstate, or
Number of Wholesale
as an inspector with the Board? obtain any license through the Board.
Distributors
1 When I first started as an inspector, the
2019 version of US Pharmacopeia (USP)
General Chapter was incorporated by
4 | JANUARY 2022R E C E N T R E G U L AT O R Y
T R E N D S R E F L EC T
E-PRESCRIBING’S
IMPACT
on Curbing
Opioid Epidemic
efore Drug Enforcement Administration (DEA) published an
B interim final rule in 2010, which gave practitioners the option
to write prescriptions for controlled substances (CS)
electronically, e-prescribing for CS was prohibited in many states.
Now, 11 years later, the regulatory and technological landscape has
shifted significantly.
As part of the larger effort to curb the opioid crisis, many states are E-Prescribing Has Become Widely Utilized
now (or will soon be) mandating e-prescriptions for certain CS. This E-prescribing is the practice of using digital methods to transmit
shift in the general perception of e-prescribing – from vulnerability prescription information between a prescriber and the dispensing
to asset in the opioid crisis – means that even jurisdictions that are pharmacy. The more traditional handwritten prescriptions
not currently requiring e-prescribing for certain types of CS may be continue to be utilized but have started to be seen as less secure
considering such legislation in the near future. and more prone to potential abuse, in part, because abusers and
JANUARY 2022 | 5drug traffickers have grown more sophisticated in their use of
technology that makes it easier to defeat watermarks and other
security measures used with paper prescriptions. Meanwhile, . . . security protocols and
security protocols and procedures for e-prescribing have improved
and are often integrated directly into providers’ electronic procedures for e-prescribing
health record systems, making this a more secure option.
From a regulatory standpoint, e-prescribing has two major
have improved and are often
benefits that are often cited. First, the records and security integrated directly into
protocols that accompany e-prescribing generally make it
harder for those who abuse prescription opioids to receive providers’ electronic health
them. This vigilance to protect the prescription drug supply record systems, making
during the ongoing opioid crisis remains important, particularly
given the recent increase in overall opioid overdoses (see the this a more secure option.
October 2021 issue of Innovations for more details).
Another benefit of e-prescribing is a reduction in medication
errors. With handwritten prescriptions, there is more room for error
from both the dispenser and the prescriber. For example, handwriting
can more easily be misinterpreted by a pharmacist as compared to to the NABP 2022 Survey of Pharmacy Law, at least 22 states
type. Also, studies have shown that a prescriber is more likely to and jurisdictions require e-prescribing for certain drugs. More
make a mistake when handwriting a prescription in the first place. information gathered by MDToolbox, a company that offers
E-prescribing software can prevent some of these mistakes and also e-prescribing software, indicates that, when factoring in states
removes several of the steps involved in filling a prescription. with “pending legislation,” at least 37 states will mandate
In fact, some of these regulatory changes have already occurred e-prescribing in some form within the next few years.
at the federal level. In 2018, Congress passed the Substance Use- These laws have significant variations in their approach to
Disorder Prevention that Promotes Opioid Recovery and Treatment enforcement methods, waivers, and exemptions, and which
for Patients and Communities Act. Among the law’s provisions is a medications require e-prescribing.
requirement for Medicare Part D or Medicare Advantage prescription Regarding which drugs are required to be prescribed electronically,
drug plans to begin requiring e-prescribing for Schedule II-V there appear to be three major categories of law. The first is
CS. As of press time, Centers for Medicare & Medicaid Services comprised of laws that require all prescription medications to
(CMS) has set the enforcement start date for these requirements for be submitted electronically. For example, in Florida, House Bill
January 1, 2023. Food and Drug Administration (FDA) has also 831 requires prescribers to generate and transmit all prescriptions
delayed the start date for compliance action for Part D prescriptions electronically, except under certain conditions, such as conflict
written for beneficiaries in long-term care facilities to January 1, 2025. with FDA restrictions on e-prescribing or prescriptions issued to
individuals receiving hospice care or who are in an assisted living
Majority of States Now Mandate E-Prescribing facility. A waiver process is available to prescribers who meet certain
for at Least Some Medications conditions.
At the state level, the trend has been dramatically shifting According to the MDToolbox data, 18 states’ e-prescribing
toward e-prescribing requirements in recent years. According requirements are limited to CS. An additional eight states have more
specific requirements under which drugs are required to be
electronically prescribed. Some states, such as Maine and Virginia,
are applying their e-prescribing mandates only to medications
. . . when factoring in states that contain opioids. Others are limiting the requirement to apply
only to certain schedules of CS. For example, Arizona’s laws require
with “pending legislation,” at Schedule II CS to be electronically prescribed, while Colorado’s law
includes Schedule II-IV CS.
least 37 states will mandate It should be noted that during the coronavirus disease 2019
e-prescribing in some form pandemic, some states waived or postponed implementation of their
e-prescribing requirements to make it easier for health care providers
within the next few years. to provide phone-in and written prescriptions when needed.
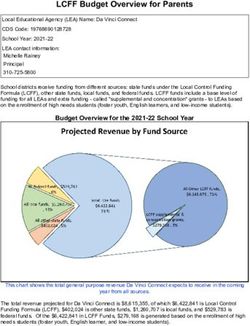
6 | JANUARY 2022State Overview of
E-Prescribing Requirements
States mandating States mandating States with
e-prescribing for e-prescribing for more limited
all prescriptions (9): all controlled requirements (8):
substances (18):
CA NV AR (Schedule II-VI CS)
FL NY CT NM AZ (Schedule II CS)
IA PA IL OK CO (Schedule II-IV CS)
MI WV IN RI KS (all CS containing
MN KY SC opioids)
MA TN ME (all CS containing
MO TX opioids)
NE UT MD (Schedule II)
NH WA
NJ WY NC (certain Schedule II
and Schedule III CS)
VA (all prescriptions
containing opioids)
NABP Resolutions on E-Prescribing
NABP has been closely watching e-prescribing laws and regulations • Resolution 115-1-19 resolves that NABP should engage
for some time. Recent action related to the practice has been the stakeholders to encourage prescribers and pharmacists to use
subject of two Annual Meeting resolutions from 2018 and 2019. e-prescribing transactions to avoid duplicative or inappropriate
prescribing and medication therapy.
• Resolution 114-3-18 acknowledges evidence that mandating
e-prescribing provides multiple advantages and resolves that E-prescribing is just one of many tools that health care providers and
NABP collaborate with appropriate stakeholders, including DEA, regulators are utilizing in their efforts to curb prescription drug misuse
CMS, and e-prescribing experts, to examine the feasibility of and abuse. NABP will continue to monitor changes in e-prescribing
mandating that all prescriptions be transmitted electronically. regulations and their effect on pharmacy practice and regulation.
Further updates will be provided in future communications.
JANUARY 2022 | 7ASSOCIATION NEWS
FDA Delays Enforcement Date for Statutory Compounding
Limit, Extends Deadline for States’ MOU Decisions
In summer 2021, Food and Drug will not have a significant economic effect such products interstate and report those
Administration (FDA) announced that on small businesses or prepare a regulatory pharmacies to FDA. Boards can use the
states would receive an additional 12 flexibility analysis. information sharing network, accessible
months to decide whether to sign its The FDA Compounding MOU Project via NABP e-Profile Connect, to meet the
Memorandum of Understanding Addressing was established through a partnership obligations outlined in the MOU. While
Certain Distributions of Compounded between NABP and FDA and aims boards are not required to enter data into
Human Drug Products (MOU) before to improve data sharing related to the network, they are encouraged to do so to
the agency would enforce its statutory 5% compounding pharmacies. As part of the create a uniform and streamlined reporting
limit on out-of-state distribution. This project, NABP developed the information process with FDA.
announcement came following a request sharing network to help state boards of
from some states and NABP for additional pharmacy collect, manage, and share data Truths and Misconceptions
time to determine the legal and logistical related to compounding pharmacies with About the MOU
ramifications of signing the MOU. Per FDA in order to meet the obligations of the NABP has identified a few misconceptions
the announcement, enforcement of the MOU. FDA worked with NABP to develop that have circulated among boards
statutory limit will now begin on October a standard MOU for use by the state boards of pharmacy and stakeholders. These
27, 2022. At that time, the 5% limit will of pharmacy to aid their compliance with misconceptions can impede the enactment
only apply to pharmacies in states that have section 503A(b)(3)(B)(i) of the Federal of this measure that the Association believes
not signed the MOU. Food, Drug, and Cosmetic Act. States that is vital to the protection of public safety.
As of press time, FDA is evaluating a sign the MOU must identify pharmacies The first of these misconceptions is that
September 2021 court order that remanded that are compounding human drug products states can negotiate their own versions of
the MOU to FDA to either certify that it and distributing inordinate amounts of the MOU. Understandably, the standard
8 | JANUARY 2022ASSOCIATION NEWS
MOU is meant to avoid a patchwork of compounded human drug products shipped
agreements that FDA and pharmacies out of state by compounding pharmacies.
located in multiple states would need to Finally, another misconception relates By signing the MOU and
track and comply with. There is only one to inspections and investigations of participating in the project,
MOU that exists, and all states that sign compounding physicians’ offices. The boards of pharmacy
it will be under the same obligations. MOU does not require boards to enter are obligated to report
Another misconception is that the physicians’ offices to inspect or investigate the following to FDA:
MOU forces states to scrutinize every compounding activities. It only requires
compounded prescription. States do states to report complaints of adverse • harmacies that are
P
not need to evaluate and verify every drug events or product quality issues compounding human drug
products and distributing
compounded drug prescription to determine for compounded human drug products
inordinate amounts
whether a pharmacy has met the inordinate compounded by physicians’ offices and
interstate, as well as certain
amount threshold. The information shipped out of state, if they become aware compounding data.
sharing network was created specifically of such complaints. In addition, the MOU
to help states more easily determine and requires states to report if they become • omplaints of serious
C
adverse experiences or
report this information. States may also aware that a physician’s office is shipping any
quality issues relating to
utilize the information sharing network amount of compounded products interstate.
human drug products
to report complaints of serious adverse Greater compounding oversight compounded by pharmacies
drug events and product quality issues for reduces the chance of another tragedy and distributed interstate.
like the 2012 multistate outbreak of
fungal meningitis, which was linked to
• omplaints of adverse
C
experiences or quality issues
drug products compounded by the New relating to human drug
England Compounding Center (additional products compounded
Boards can use the information on the latest regulatory by a physician and
information sharing responses to the tragedy is available in distributed interstate.
network, accessible via the November/December 2021 issue of
• I nformation relating to
Innovations). Increased oversight provided the interstate distribution
NABP e-Profile Connect, by the MOU also helps ensure greater of any amount of human
to meet the obligations patient confidence that regulators are drug products compounded
outlined in the MOU. closely watching the facilities that produce by physicians.
compounded drugs that improve and
save lives.
Volunteer to Serve on a Committee or Task Force
NABP is seeking volunteers from its encouraged to submit an application Members section of the NABP website
active member boards of pharmacy to and an up-to-date résumé or curriculum under Board Resources.
serve on its 2022-2023 committees vitae. Board of pharmacy staff interested All materials will be forwarded to
and task forces. Executive officers and in volunteering for NABP task forces NABP President-elect Reginald B.
current board members, including are also encouraged to apply. “Reggie” Dilliard, DPh, who will make
public members, interested in serving Please apply online by Friday, June the appointments following the 118th
on a committee or task force are 3, 2022. The form is available in the NABP Annual Meeting.
JANUARY 2022 | 9ASSOCIATION NEWS
NABP Clearinghouse 2021
3rd
Quarter Over 1,400 Disciplinary Actions Recorded
1st Quarter 2nd Quarter 4th Quarter
Of the 1,425 actions reported in third quarter 2021:
• 514 (36%) were on pharmacists;
The Association’s data results for
• 475 (33%) were on pharmacies;
the third quarter of 2021 showed • 326 (23%) were on pharmacy technicians;
that a total of 1,425 disciplinary • 38 (3%) were on wholesalers, manufacturers, and distributors;
records were submitted to the • 32 (2.2%) were on other individuals;
NABP Clearinghouse by state boards • 20 (1.4%) were on pharmacy interns;
of pharmacy on 1,236 individual • 12 (0.8%) were on other licensees;
and business NABP e-Profiles. • 4 (0.3%) were on Drug Enforcement Administration and
Food and Drug Administration registrations; and
• 4 (0.3%) were on controlled substance licenses.
Third Quarter 2021 Action Code Categories INDIVIDUALS Third Quarter 2021 Bases for Action Code Categories INDIVIDUALS
COUNT % COUNT % COUNT % COUNT %
Publicly Available Fine/ License/Certificate
256 23.1% Restored or Reinstated, Noncompliance With Fraud, Deception, or
Monetary Penalty 82 7.4% 462 45.1% 39 3.8%
Complete, Conditional, Requirements Misrepresentation
Partial, or Denied
Probation of License 137 12.3%
Reduction, Modification, Improper Prescribing,
or Extension of Previous 44 4% Dispensing, Improper Supervision
Other Licensure Actions - Licensure Action Administering 196 19.1% 31 3%
122 11% or Allowing Unlicensed
Not Classified Medication/Drug Practice
Summary or Emergency Violation
Revocation of License/ Action, Limitation,
100 9% 34 3.1%
Certificate Suspension, or Restriction
on License Other Licensure Actions -
121 11.8%
Suspension of License/ Not Classified Misconduct or Abuse 18 1.8%
100 9%
Certificate Limitation or Restriction on
17 1.5%
License Criminal Conviction or
87 8.5%
Voluntary Surrender of Adjudication
100 9%
License/Certificate Denial of Initial License or
14 1.3% Confidentiality, Consent,
Renewal License/Certificate 5 0.5%
Unsafe Practice or or Disclosure Violation
66 6.4%
Reprimand or Censure 96 8.6% Substandard Care
Miscellaneous 8 0.7%
TOTAL 1,110 TOTAL 1,025
Third Quarter 2021 Action Code Categories BUSINESSES Third Quarter 2021 Bases for Action Code Categories BUSINESSES
COUNT % COUNT % COUNT % COUNT %
Publicly Available Fine/ License/Certificate Restored Fraud, Deception, or
407 54% Noncompliance With 22 2.8%
Monetary Penalty or Reinstated, Complete, 491 63.4% Misrepresentation
4 0.5% Requirements
Conditional, Partial, or
Denied
Reprimand or Censure 241 32%
Other Actions - Not
13 1.7%
Monitoring, Closure, or Classified
Probation of License 34 4.5% Other Operational Business 4 0.5% Improper Supervision
Modification or Allowing Unlicensed 202 26.1%
Voluntary Surrender of Practice
21 2.8%
License/Certificate Reduction, Modification, Confidentiality, Consent,
6 0.8%
or Extension of Previous 1 0.1% or Disclosure Violations
Revocation of License/ Licensure Action
19 2.5% Improper Prescribing,
Certificate
Dispensing,
Suspension of License/ Administering 38 4.9% Criminal Conviction or
Other Licensure Actions - 7 0.9% Medication/Drug 2 0.3%
16 2.1% Certificate Adjudication
Not Classified Violation
TOTAL 754 TOTAL 774
10 | JANUARY 2022118th ANNUAL MEETING
Join Fellow NABP Members in the Valley of the Sun for the
118th Annual Meeting
NABP invites its members and other Specifically, the Annual Meeting
pharmacy stakeholders to Phoenix, allocates time for board delegates to elect
AZ, for the Association’s 118th Annual new Executive Committee officers and
Meeting. Themed “Expanding Our members, discuss proposed amendments Online Registration Will
Vision to Advance Public Health to the NABP Constitution and Bylaws, Soon Be Available on the
Protection,” the Annual Meeting will be and vote on proposed Association Annual Meeting Website
held May 19-21, 2022, at the Sheraton resolutions. In addition, ample sessions In February 2022, check out
Grand at Wild Horse Pass. Join your and events provide attendees with the the Annual Meeting website
regulatory colleagues for important opportunity to participate in continuing for the latest information about
Association business sessions, education, pharmacy education activities and to the 118th Annual Meeting.
and networking opportunities. network with peers. Online registration, hotel and
transportation details, and
more will soon be available at
E NABPAnnualMeeting.pharmacy
N
O
X
O
L
A
N
Travel Grant Available to Attend 118th
NABP Annual Meeting in Phoenix
The NABP Foundation® is once again offering travel grant opportunities for
individuals planning to attend the 118th NABP Annual Meeting in Phoenix,
AZ. Eligible individuals may receive up to $1,500 to cover the cost of travel,
hotel rooms, meals, taxis, parking, and tips. The grant does not include
registration fees. All applicants will be informed of whether they have
qualified for the grant.
• One grant will be awarded to a current board member or administrative
officer of each active NABP member board of pharmacy, as designated
by the board’s administrative officer.
• Active member boards of pharmacy must have a voting delegate in
attendance at the Annual Meeting to vote during all applicable business
sessions in order to receive reimbursement.
To obtain a grant application, board administrative officers may contact
ExecOffice@nabp.pharmacy.
JANUARY 2022 | 11118th ANNUAL MEETING
Proposed Resolutions Will Be Distributed in February
Proposed resolutions received at NABP be received by Friday, April 29, 2022, in
Headquarters by Friday, February 11, accordance with Article IV, Section 6, Part
2022, will be distributed electronically to (d) of the NABP Constitution and Bylaws. Important Deadlines
state boards of pharmacy on the following Resolutions not submitted at least 20 days
• ebruary 11, 2022
F
Thursday, February 17, 2022, for review prior prior to the Annual Meeting, but submitted Proposed resolutions
to the 118th NABP Annual Meeting. This within a time frame that the Executive must be received at
mailing will constitute the only preconference Committee deems appropriate (prior to the NABP Headquarters for
distribution of proposed resolutions. All meeting of the Committee on Resolutions), preconference distribution to
resolutions – those distributed for early may be presented during the Annual Meeting the state boards of pharmacy.
review as well as those received after February and will be considered for adoption by the
• ebruary 17, 2022
F
11 – will be presented to the voting delegates Association upon the affirmative vote of Proposed resolutions are
during the Second Business Session of three-fourths (3/4) of those active member distributed electronically to
the Annual Meeting by the chair of the boards present and constituting a quorum. state boards of pharmacy
Committee on Resolutions and subsequently Questions regarding resolution for review.
voted on during the Final Business Session. procedures should be directed to the
• pril 29, 2022
A
Any active member board, district, or NABP Executive Office via email at Proposed resolutions must
committee of the Association may submit ExecOffice@nabp.pharmacy. be submitted to be considered
E X PA
resolutions to NABP. To be considered N at the Annual Meeting.
during the Annual Meeting, resolutions must
DI
NG VISIO
N
Submit Your Poster Proposal by February 23
Limited Spots Available – 2022, at the 118th NABP Annual preparing for the North American
Don’t Delay! Meeting in Phoenix, AZ. To be Pharmacist Licensure Examination.
NABP is seeking proposals for its considered for the Poster Session, Poster Session presenters may be
annual Educational Poster Session. individuals must be able to attend eligible to earn Accreditation Council
Board of pharmacy members and the in-person meeting on May 21. for Pharmacy Education-accredited
staff, as well as schools and colleges of Selected poster presenters must also continuing pharmacy education
pharmacy, are invited to submit their be available in March and April for credit. Details will be provided
proposals as they relate to this year’s correspondence with NABP staff and to individuals who are selected
poster session theme of “Sharing to submit required materials. to present posters. Those selected
Our Vision to Advance Public Students are welcome to submit to present a poster will receive a
Health Protection.” Poster proposals poster proposals. If selected, the complimentary meeting registration.
may be descriptive, scientific, or student(s) must be accompanied by Interested in submitting
informational in nature. Possible a credentialed advisor or licensed a proposal? Contact NABP
topics include policy development, pharmacist. All participating Professional Affairs staff
public health initiatives, and pharmacy school students will via email at Prof-Affairs@
legislative issues, among others. receive a complimentary voucher nabp.pharmacy for detailed
The Poster Session will be held in their NABP e-Profile valued at instructions. Proposals must
the morning of Saturday, May 21, $75 to take the Pre-NAPLEX®, a be submitted by Wednesday,
practice examination for students February 23, 2022.
12 | JANUARY 2022118th ANNUAL MEETING
NABP Announces 2022-2023 Executive
Committee Openings; Elections to Take Place
During Annual Meeting
As of press time, NABP has received the following nominations for the open Executive Committee officer
and member positions:
Expand Vision
President-elect (one-year term) District 1 (three-year term) District 3 (one-year term)
Lenora S. Newsome, PD, Bradley S. Hamilton, BSPharm, Traci Collier, PharmD, RPh,
Arkansas RPh, Maine South Carolina
Treasurer (one-year term) District 2 (three-year term) District 5 (three-year term)
Jeffrey J. Mesaros, PharmD, JD, Janet Getzey Hart, RPh, Shane R. Wendel, PharmD, RPh,
RPh, Florida Pennsylvania North Dakota
Updates to the list of nominations will be posted on the Annual Meeting page in the About section of www.nabp.pharmacy.
Individuals interested in running for an open officer or member position must submit a letter of intent, including the
expiration date for their term on the active member board, and a résumé or curriculum vitae to the NABP executive
director/secretary at least 45 prior (by April 4, 2022) to the Annual Meeting’s First Business Session.
Executive Committee Nomination and Election Process
NABP/AACP District Meetings
Candidate
Qualifications
Members are nominated by the district to run for the open
Executive Committee member positions for their district.* • ust be an affiliated
M
member (administrative
officer or board member)
of the Association currently
Annual Meeting
serving on a board of
First Business Session pharmacy of an active
Candidates for open Executive Committee member and officer positions introduced. member state at the time of
nomination and election
• ust not currently serve as
M
an officer, official, or board
Second Business Session or staff member for any
national or state pharmacy
Candidate and seconding speeches are presented.
organization
• ust not have a conflict of
M
interest with the purpose,
mission statement, and
Final Business Session
operation of NABP
Board of pharmacy delegates vote for new Executive Committee
members and officers on behalf of their board. Newly elected officers More information about the
and members are installed during the Final Business Session. procedures for nominating and
electing Executive Committee
officers and members is
*Individuals may submit their nomination outside the district process for the open member
available in Article IV, Sections
positions. Only those individuals who have been determined by NABP to meet all qualifications
3(b) and 3(c) of the NABP
for the open member positions will be placed on the ballot. More information can be found in the
Constitution and Bylaws.
NABP Constitution and Bylaws, which can be accessed in the About section of the NABP website.
JANUARY 2022 | 13INTERVIEW WITH A BOARD MEMBER
Ashlee Bow, PharmD, RPh, AAHIVP
Member, District of Columbia Board of Pharmacy
When were you appointed to Has the Board encountered any
the Board of Pharmacy? Are you challenges to developing and/or
a pharmacist, technician, public implementing these new policies,
member, or other type of member? legislation, or regulations?
I have served as a pharmacist member of the There are always challenges that arise
District of Columbia Board of Pharmacy when changes are being made. The
since March 2019. I have been a pharmacist coronavirus disease 2019 pandemic has
for eight years, working in a community been a major challenge for us, forcing
pharmacy, and have worked in the District us to put some things on hold while we
of Columbia since 2016. I currently serve as worked to create emergency regulations.
chair of the Communications Subcommittee All of our meetings are now virtual, and
and am a member of the Pharmacy Laws and pharmacists are really busy during this time,
District of Columbia Regulations Subcommittee. so scheduling can also be a challenge.
Board of Pharmacy
What steps should a board member What advice would you give to a new
Number of Board take to be successful in their role? Board member?
Members Doing your homework before board New board members should have the
5 pharmacist members meetings is the most important step in confidence to ask questions when they do
and 2 public members being a successful board member. Before not understand something or need more
each meeting, I go through all of the clarification. It is important to stay informed
meeting materials and often research and up to date as much as possible on
Number of Compliance what is happening in other states so that current events in pharmacy and to have a
Officers/Inspectors I can fully understand any issues we may working knowledge of the regulations in
6 discuss. I also find myself constantly using your jurisdiction. It is very important that
NABPLAW® Online, depending on what we new members understand the commitment
are discussing during our meetings. You can it takes to be on a board of pharmacy and
Rules & Regulations
never be too prepared for a meeting. the time that they will need to invest.
Established by
District of Columbia What are some recent policies, Have you served as a member of any
Board of Pharmacy legislation, or regulations that NABP task forces or committees, or
and the District of your Board has implemented or is attended NABP or district meetings?
Columbia mayor
currently working on? If so, in your experience, what are
The Board’s Laws and Regulations the benefits of participating in these
Subcommittee has been working diligently to NABP activities?
Number of
update regulations. We have been reviewing I am not currently serving on an NABP
Pharmacist Licensees
2,036 each chapter of the District of Columbia’s task force, but I attended the virtual 117th
Pharmacy Laws and Regulations, which NABP Annual Meeting in May 2021. It
include pharmacist-in-charge requirements, was very refreshing to hear new ideas from
immunizations, 90-day refills, and tech- other board members, and there were great
Number of check-tech. We are also putting together a networking opportunities to meet members
Pharmacies
workplace survey for pharmacists, pharmacy from other states. I particularly enjoyed the
178
technicians, pharmacy technician trainees, breakout rooms, as I was able to engage with
and pharmacy interns in the District. other attendees on familiar topics in which
I had some interest as well as gain exposure
Number of Wholesale to new topics.
Distributors
27
14 | JANUARY 2022AROUND THE ASSOCIATION
Executive Officer Changes
• J ames R. Skizewski has been named • ark Bunton, RPh, has been
M • heryl Lynn “Cheri” Garvin, RPh,
C
executive officer of the Hawaii State appointed a member of the Indiana has been appointed a member of the
Board of Pharmacy, replacing Lee Board of Pharmacy. Bunton’s Virginia Board of Pharmacy. Garvin’s
Ann Teshima. Skizewski also serves as appointment will expire October appointment will expire June 30, 2025.
executive officer for the Hawaii Board 1, 2024.
of Elevator Maintenance and the • J ason Jablonski, RPh, has been Board Member Reappointments
Certified Nursing Aide and Nursing
Home Administrator programs. He
appointed a member of the Indiana • J onathan Brunswig, PharmD, RPh,
Board of Pharmacy. Jablonski’s has been reappointed a member
graduated from the University of appointment will expire October of the Kansas State Board of
Hawaii at Manoa with a bachelor’s 5, 2024. Pharmacy. Brunswig’s appointment
degree in governance and sociology.
• J ames “Jim” Mennen, RPh, has been will expire April 30, 2025.
• hristine M. Horne has been
C appointed a member of the Iowa Board • ill Walden, RPh, has been
B
named board administrator III of Pharmacy. Mennen’s appointment reappointed a member of the
of the New Hampshire Board of will expire April 30, 2024. Kansas State Board of Pharmacy.
Pharmacy, replacing Traci Weber.
She has worked for the state of • ucinda Noches Talbert has
L Walden’s appointment will
been appointed a public member expire April 30, 2025.
New Hampshire for eight years and
is currently board administrator
of the Kansas State Board of • yle A. McCree has been reappointed
K
Pharmacy. Talbert’s appointment a public member of the Michigan
III for nine boards. Horne holds
will expire April 30, 2025. Board of Pharmacy. McCree’s
an associate’s degree in business
management from Hesser College. • ierre Boutros, RPh, has been
P appointment will expire June 30, 2025.
• Brad Wojciechowski has been named
appointed a member of the Michigan • J illian Foster, MBA, PharmD, has
Board of Pharmacy. Boutros’ been reappointed a member of the
executive director of the Wisconsin
appointment will expire June 30, 2025. Mississippi Board of Pharmacy. Foster’s
Pharmacy Examining Board, replacing
Christine Poleski. Wojciechowski • arolyn R. Bodell, RPh, has
C appointment will expire June 30, 2026.
graduated from the University been appointed a member of the • yan Harper, PharmD, RPh, has
R
of Wisconsin-Eau Claire with a North Dakota State Board of been reappointed a member of the
bachelor’s degree in political science. Pharmacy. Bodell’s appointment Mississippi Board of Pharmacy.
will expire May 8, 2026. Harper’s appointment will expire
Board Member Appointments • on J. Horner has been appointed a
R June 30, 2026.
• atrick Adams, RPh, has been
P public member of the North Dakota • enneth Kenyon, PharmD, RPh,
K
appointed a member of the Hawaii State Board of Pharmacy. Horner’s BCPS, has been reappointed a
State Board of Pharmacy. Adams’ appointment will expire May 8, 2026. member of the Washington State
appointment will expire June 30, 2023. • J ason George, PharmD, RPh, has Pharmacy Quality Assurance
been appointed a member of the State Commission. Kenyon’s appointment
• ark Brown, RPh, has been
M
will expire January 19, 2025.
appointed a member of the Hawaii of Ohio Board of Pharmacy. George’s
State Board of Pharmacy. Brown’s appointment will expire June 30, 2025. • oang-Uyen Thorstensen, CPhT,
H
appointment will expire June 30, 2025. • od Joseph “T.J.” Grimm, MBA,
T has been reappointed a member of
RPh, has been appointed a member of the Washington State Pharmacy
• atalina Cross, PhD, has been
C
Quality Assurance Commission.
appointed a public member of the the State of Ohio Board of Pharmacy.
Grimm’s appointment will expire June Thorstensen’s appointment will
Hawaii State Board of Pharmacy. Cross’
30, 2024. expire January 19, 2025.
appointment will expire June 30, 2023.
• ent Kikuchi, MBA, RPh, has been
K • ichard Joyce, CPhT, has been
R
appointed a member of the Hawaii appointed a member of the Oregon
State Board of Pharmacy. Kikuchi’s State Board of Pharmacy. Joyce’s
appointment will expire June 30, 2025. appointment will expire February
29, 2024.
• heri M. Tokumaru, PharmD,
S
BCCCP, has been appointed a • dward G. Misto, RPh, has
E
been appointed a member of the
member of the Hawaii State Board of
Rhode Island Board of Pharmacy.
Pharmacy. Tokumaru’s appointment
Misto’s appointment will expire
will expire June 30, 2024.
September 1, 2024.
JANUARY 2022 | 15STATE BOARD NEWS
Kansas Pharmacist
Education Program to Focus
on PDMP Utilization
In fall 2021, the Kansas prescription drug
monitoring program (PDMP), K-TRACS,
began outreach to Kansas pharmacies to
gauge interest in peer-to-peer education
regarding K-TRACS utilization. Also
known as academic detailing, the program
is intended to help pharmacists incorporate
K-TRACS into their clinical workflows and
adopt best practices to prioritize patient • rior to renewing or applying for
P disease 2019 vaccine to attend
safety. The K-TRACS staff pharmacist licensure in Tennessee, an out-of-state K-12 schools.
will meet with peers to identify challenges pharmacy practice site must submit to More information is available in the
in their pharmacy related to controlled the Board its most recent inspection by Board’s September 2021 Newsletter.
substance (CS) dispensing and K-TRACS the regulatory agency of its respective
usage. They will then work together to state, conducted within the past year. Wyoming Implements New Sterile
identify and implement noncommercial, • pon learning that a health care
U Compounding Inspection Process
evidence-based solutions that promote prescriber was indicted of certain A new sterile compounding inspection
positive patient outcomes. criminal offenses (CS violations or sexual process will be implemented in Wyoming
More information is available in the offenses), the Tennessee Department starting this current fiscal year (July 1,
Board’s September 2021 Newsletter. of Health’s licensing authorities are 2021-June 30, 2022). All sterile
required to automatically restrict compounding inspections will now be
the prescriber’s ability to prescribe
New Mexico Approves HIV completed in two phases. The first phase
Schedule II CS until the case reaches a
Post-Exposure Prophylaxis and will consist of a virtual pre-inspection,
final disposition. The restriction shall
Point-of-Care Testing be removed upon sufficient proof of and the second phase will include an
The New Mexico Board of Pharmacy acquittal or dismissal/nolle prosequi. unannounced on-site inspection. The
approved pharmacist prescriptive authority Further, licensing authorities are virtual pre-inspection will consist of a
for the prescribing of HIV post-exposure required to automatically revoke the comprehensive review of policies, records,
prophylaxis therapy in conjunction with license of a practitioner who is convicted and documentation. All pharmacists-in-
point-of-care testing. Pharmacists who wish of those same criminal offenses. charge will be notified of the virtual
to obtain this prescriptive authority must • egulatory changes prohibit Governor
R pre-inspection and provided 30 days
go through Board-approved training and Bill Lee from issuing an executive order to compile and submit all required
adhere to the Board-approved protocol. and a state agency, department, or information. The intent of the virtual
Portions of this training that are in addition political subdivision from promulgating, pre-inspection is to prevent interruptions
to the required Accreditation Council adopting, or enforcing an ordinance in daily operations that typically occur
for Pharmacy Education-accredited or resolution that requires a person to while reviewing such records on site. Once
training for prescriptive authority are receive an immunization, vaccination, the virtual pre-inspection is complete
approved as acceptable toward the 30- or injection for the SARS-CoV-2 virus and reviewed with the designated person,
hour continuing pharmacy education or any variant of the SARS-CoV-2 virus. inspectors will follow up with a random,
requirement outlined in 16.19.4.10 In addition, new regulation deletes the unannounced on-site inspection. This
New Mexico Administrative Code. previous override during an epidemic or second phase will consist of completing the
immediate threat of an epidemic of an
associated retail or institutional inspection
objection against vaccination that was
Tennessee Legislative Updates checklists and observation of processes,
made based on religious tenets, and
Address Compounding, Criminal prohibits requiring the coronavirus
techniques, and adherence to procedures.
History, and Immunizations
The Tennessee Board of Pharmacy
implemented several legislative
updates related to the regulation of Most articles published in State Board News are selected
compounding pharmacies, criminal from the newsletters of state boards that participate in
history background, and immunizations. the NABP State Newsletter Program. Issues are posted on
The following is a summary of these the NABP website on each participating state’s page.
changes that are now in effect.
16 | JANUARY 2022You can also read